VACUODERMIE – PRESSOTHERAPY (THE MEDICAL ASPECT)
VACUODERMIE
This technique is based on the multisecular principle of vacuum used in medicine, and on the massage indications, practice and results.
The tissue vacuodermie provides a different approach to daily health care and open the way to considerable possibilities for all treatments involving connective tissue dermis, muscles, tendons, nerves etc…
Being able to choose between stationary or sliding techniques with a selection of transparent cups brings versatility and precision in treating different pathologies.
PRESSOTHERAPY
In 1984, Eureduc, in collaboration with health care specialists, invents a computer controlled compression device designed to promote venous and lymphatic flow.
The automatically modulated double gradient sequential system inflates individual sections of a chambered garment positioned around the limbs.
Pressotherapy works by progressively moving extra cellular fluid in a distal to proximal direction making it the mean of choice to treat circulatory and lymphatic condition, and lymphoedema related ailments.
VACUODERMIE – PRESSOTHERAPY (FOR THE PRACTICIAN)
For the family practitioner (M.D.), Gynecologist-obstetrician, podiatrician, oncologist, phlebologist, rheumatologist, dermatologist and lymphoedema specialist, Vacuomobilization and Pressotherapy equipment is utilized in a wide range of non-invasive treatments or therapies such as:
- alleviating leg discomfort and edema by addressing the venous return insufficiency
- micro-circulation thru vacuodermie
- improving wound healing time and scar appearance
- pre-post oncologic treatment to prevent or to reduce lymphoedema
- reeducation of the lymphatic system
It’s also useful in the treatment of Neuro-vegetative syndromes, Headache, Fibromyalgia, Neurology (hemiplegia), Scars, Osteoarthritis and an effective tool for managing Back pain, Neck (cervical) pain, Athletic traumatology, Muscle injuries, Tendon injuries.
In a Medi-spa environment, tissue vacuodermie and lymphatic drainage addresses the growing demand from “Baby Boomers” for the prevention, rejuvenation and maintenance strategies.
VACUODERMIE – PRESSOTHERAPY (FOR SURGERY)
In the surgery field, pressotherapy help control the vascular, venous and lymphatic systems, with applications in urology, reconstructive and plastic surgery, traumatology, orthopedic surgery, obstetrical surgery, oncology, vascular surgery.
APPLICATIONS
- primary and secondary lymphoedemas
- post-thrombosic edema, hypodermia
- post-thrombosis or venous ulcers
- post-traumatic edema
- lypodystrophies
Thrombosis
The thrombosis of the deep veins of the lower limbs is a frightening post-operative complication in many surgical acts. It often starts forming during the surgery itself because it is induced by immobility, hyper-coagulability at the time of a particularly hemorrhagic intervention, the possible mobilization of the intra-abdominal bodies with the possible compression of the deep venous axes.
To prevent thrombosis; Pressotherapy creates a true per operational assistance of the venous movement of the lower limbs by a pneumatic compression done via boots with compartments enclosing the members to be treated. This gradient of pressure allows a progressive increase in the tissue pressure (stronger partly distal than proximal) and a powerful venous drainage.
VACUODERMIE – PRESSOTHERAPY (PRE & POST SURGERY)
Vacuomobilization & Pressotherapy can be fully integrated into pre- & post-surgical procedures, helping prepare the patient for surgery, and providing a range of post-surgical treatment allowing faster recovery. It can be performed in a doctor’s office, hospital clinic or medical spa, by a licensed and trained medical assistant, nurse or an esthetician.
PRE-LIPOSUCTION
The connective tissue contains multiple vascular, lymphatic and nervous structures in direct contact with adipose tissue and fat lobes, responsible for either build up or eliminate fatty unwanted deposits.
Vacuodermie gently stretch the connective tissue allowing room for local circulation, improving metabolism oxygenation, cell rejuvenation process and elimination of toxic waste.
Vacuomobilization intensive irrigation will help brake down & soften the fibrotic tissue in preparation for liposuction, resulting in easier access to the surgical implements, fewer bruises, contusions, ecchymosis, therefore improving the healing process.
POST-LIPOSUCTION
Following evaluation and recommendation of the surgeon
Mechanical Lymphatic Drainage – Day 21 to 120 (1 x weekly)
- Pressotherapy alleviates swelling & edema
- Improves venous circulation
- Reduces sensitivity
- Diminishes bruising & hematoma
- Fights post-operative adhesion
Post-liposuction sliding vacuomolization maintenance treatments are recommended monthly to improve vascularization of the adipose tissue and activate micro-circulation. Vacuomobilization stimulates fibroblast, augmenting collagen and elastin fibers function & synthesis to help build and strengthen the tissue, increasing skin tone.
VACUODERMIE – PRESSOTHERAPY (RECONSTRUCTIVE APPLICATIONS)
The goal of vacuomobilization is to decrease the evolution of the scar through defibrosis and softening with orthodermic techniques to obtain a scar that is:
- Of better quality
- Cosmetic in appearance
- Stable within a shorter time
- Without complication
- Allowing a faster return to normal functions
Tissue vacuodermie is particularly indicated in plastic surgery, reparative or cosmetic surgery. The applied vacuum induces lifting, stretching and torsion of the scar tissue and peripheral zones with detachment of the subcutaneous conjunctive tissue vertically towards the surface, causing significant hyperemia of the treated zone.
The resulting stimulation leads to the normalization of the micro-circulation and lymphatic anastamosis with the neighboring healthy tissue. The vacuum intensity and the size of the cup can be accurately adjusted to the type of lesion to be treated.
The flexibility of the TV 20 makes it possible to treat any type of scar: normal or hypertrophied scars, burns, cutaneous lesions, surgical or traumatological.
MOTOR VEHICLE ACCIDENT
TREATMENT
30 days post injury
35 sessions – 3 x week
MODALITIES
-
Vacuomobilization
- Manual Massage
- Hydrotherapy
OUTCOME
- Symmetrical eyelid dynamics
- Flexible and flattened forehead & cheeks scars
- Significant reduction of aesthetic handicap
(Surgical revision of scar below nostril recommended)
VACUODERMIE REFERENCES
Physiotherapy of traumatic and surgical wounds
F. Souyris, J.M.Hebting, L. Montes de OCA, P. Jammet
Orbito palpebral surgery and massokinesitherapy
J.M Hebting, A. Gary-Bobo
Treating scars with masso-kinesiotherapy
J.M Hebting, G. Billotet, J.O. Bourgeois, M. Pocholle
Orthopedics and hands surgery department
Y. Allieu
The benefits of vacuomobilization is treating orthopedic scars.
J.M. Hebting, J.C. Rouzaud, A. Gary-Bobo
Physical therapy in wound care
J .M Hebting
Kinesitherapy and migraines. Illusion or realty, observations on 30 cases.
J.M Hebting, S. Lorenzo
Surgery of the thorax and physical therapy.
O. Billotet, J.M. Hebting, B. Albat
Kineplasty
J.M. Hebting, B. Allegre, O. Billotet, A. Gary-Bobo
PRESSOTHERAPY REFERENCES
Laser Doppler vasomotion among patients with post-thrombotic venous insufficiency:
Effect of intermittent pneumatic compression.
K.Pekanmaki, P.J. Kolari, U. Kistal
Treatment of lymphedema with a compression device.
Michael R. Bastian, M.D. – Beth G. Goldstein, M.D. – Jack L. Lesher, Jr., M.D. – J. Graham Smith, Jr., M.D.
Medical College of Georgia, Augusta, GA 30912-2900
Lymphoedema of the upper limb, secondary to treatment of a mammary tumour:
Statistical study of the result provided by physical treatment.
Jean Claude Ferrandez, Kinesitherapist, Avignon, Franceal
The physical treatment of post-therapeutic upper limb lymphoedema and its long-term follow up care.
J.P. Burn- Service de Reeducation Vasculaire,
Pavillon VIAT 3, Hospital Saint-Joseph – 7 rue Pierre-Larousse, F 75674 Paris Cedex 14
Interest of pneumatic compression – J.C. Ferrandez, J.P. Laroche
Prevention of Venous Thromboembolism. – Taft Academic Vascular Surgery, St. Marys Hospital Medical School
Hemodynamic alterations in venous blood flow produced by external pneumatic compression.
Herbert Janssen, Ph.D., Cristina Trevino, B.S., David Williams, M.S.
Kineplasty. Treatment of Traumatic or surgical wounds. – J.M. Hebting, B. Allegre, O. Billotet, A. Gary Bobo
Compression in the treatment of Leg Telangiectasia: Theoretical Considerations.
Mitchel P. Goldman, M.D.
The effects of the modified intermittent sequential pneumatic device (MISPD) on exercise performance following an exhaustive exercise bout.
Avigdor Zelikovski M.D., Clive L. Kaye MSc, Gershon Fink, Shimon A. Spitzer M.D., and Yair Shapiro M.D.
Prevention of Thromboses and Cutaneous Neoroses using physical methods and Pressotherapy in surgery of chronic veinous deficiency of the lower limbs.
J. Valentin, D Leonhardt, M. Perrin
Intermittent pneumatic compression
Intermittent pneumatic compression is a therapeutic technique used in medical devices that include an air pump and inflatable auxiliary sleeves, gloves or boots in a system designed to improve venous circulation in the limbs of patients who suffer edema or the risk of deep vein thrombosis (DVT) or pulmonary embolism (PE).[1]
In use, an inflatable jacket (sleeve, glove or boot) encloses the limb requiring treatment, and pressure lines are connected between the jacket and the air pump. When activated, the pump fills the air chambers of the jacket in order to pressurize the tissues in the limb, thereby forcing fluids, such as blood and lymph, out of the pressurized area. A short time later, the pressure is reduced, allowing increased blood flow back into the limb.
The primary functional aim of the device “is to squeeze blood from the underlying deep veins, which, assuming that the valves are competent, will be displaced proximally.” When the inflatable sleeves deflate, the veins will replenish with blood. The intermittent compressions of the sleeves will ensure the movement of venous blood.
Sequential compression devices
Sequential compression devices (SCD) utilize sleeves with separated areas or pockets of inflation, which works to squeeze on the appendage in a “milking action.” The most distal areas will initially inflate, and the subsequent pockets will follow in the same manner.[2]
Sequential calf compression and graduated compression stockings are currently the preferred prophylaxis in neurosurgery for the prevention of DVT and pulmonary embolism, sometimes in combination with low molecular weight heparins or unfractionated heparin.[3]
Intraoperative SCD-therapy is recommended during prolonged laparoscopic surgery to counter altered venous blood return from the lower extremities and consequent cardiac depression caused by pneumoperitoneum (inflation of the abdomen with carbon dioxide).
Lymphedema – Wikipedia
Lymphedema
Lymphedema is characterized by a retention of fluid and tissue swelling. The fluid retention and swelling is caused by a lymphatic system which has been compromised. Often times fatigue will accompany a heavy and swollen limb or an accumulation of fluid in other areas of the body, including the neck or head. If left untreated, early stages of lymphedema can cause the skin to discolor and grow in severity until deformation occurs.
| This article needs additional citations for verification. (May 2008) (Learn how and when to remove this template message) |
| Lymphedema | |
|---|---|
| lymphoedema, lymphatic obstruction | |

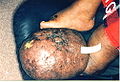
Lymphedema on a 67 year old female
|
|
| Classification and external resources | |
| Specialty | General surgery |
| ICD–10 | I89.0, I97.2, Q82.0 |
| ICD–9-CM | 457.0, 457.1, 757.0 |
| OMIM | 153100 |
| DiseasesDB | 7679 |
| eMedicine | article/1087313 |
| MeSH | D008209 |
Lymphedema is a condition of localized fluid retention and tissue swelling caused by a compromised lymphatic system, which normally returns interstitial fluid to the thoracic duct, then the bloodstream. The condition can be inherited or can be caused by a birth defect, though it is frequently caused by cancer treatments and by parasitic infections. Though incurable and progressive, a number of treatments can ameliorate symptoms. Tissues with lymphedema are at high risk of infection.
Signs and symptoms[edit]
Video explanation
Symptoms may include a feeling of heaviness or fullness, edema, and (occasionally) aching pain in the affected area. In advanced lymphedema, there may be the presence of skin changes such as discoloration, verrucous (wart-like) hyperplasia, hyperkeratosis and papillomatosis; and eventually deformity (elephantiasis).
Lymphedema should not be confused with edema arising from venous insufficiency, which is not lymphedema. However, untreated venous insufficiency can progress into a combined venous/lymphatic disorder which is treated the same way as lymphedema.
Presented here is an extreme case of severe unilateral hereditary lymphedema which had been present for 25 years without treatment:
Causes[edit]
Lymphedema affects approximately 140 million people worldwide.[1]
Lymphedema may be inherited (primary) or caused by injury to the lymphatic vessels (secondary). It is most frequently seen after lymph node dissection, surgery and/or radiation therapy, in which damage to the lymphatic system is caused during the treatment of cancer, most notably breast cancer. In many patients with cancer, this condition does not develop until months or even years after therapy has concluded. Lymphedema may also be associated with accidents or certain diseases or problems that may inhibit the lymphatic system from functioning properly. In tropical areas of the world, a common cause of secondary lymphedema is filariasis, a parasitic infection. It can also be caused by a compromising of the lymphatic system resulting from cellulitis.
While the exact cause of primary lymphedema is still unknown, it generally occurs due to poorly developed or missing lymph nodes and/or channels in the body. Lymphedema may be present at birth, develop at the onset of puberty (praecox), or not become apparent for many years into adulthood (tarda). In men, lower-limb primary lymphedema is most common, occurring in one or both legs. Some cases of lymphedema may be associated with other vascular abnormalities.
Secondary lymphedema affects both men and women. In women, it is most prevalent in the upper limbs after breast cancer surgery, in particular after axillary lymph node dissection,[2] occurring in the arm on the side of the body in which the surgery is performed. In Western countries, secondary lymphedema is most commonly due to cancer treatment.[1] Between 38 and 89% of breast cancer patients suffer from lymphedema due to axillary lymph node dissection and/or radiation.[1][3][4] Unilateral lymphedema occurs in up to 41% of patients after gynecologic cancer.[1][5] For men, a 5-66% incidence of lymphedema has been reported in patients treated with incidence depending on whether staging or radical removal of lymph glands was done in addition to radiotherapy.[1][6][7]
Head and neck lymphedema can be caused by surgery or radiation therapy for tongue or throat cancer. It may also occur in the lower limbs or groin after surgery for colon, ovarian or uterine cancer, in which removal of lymph nodes or radiation therapy is required. Surgery or treatment for prostate, colon and testicular cancers may result in secondary lymphedema, particularly when lymph nodes have been removed or damaged.
The onset of secondary lymphedema in patients who have had cancer surgery has also been linked to aircraft flight (likely due to decreased cabin pressure or relative immobility). For cancer survivors, therefore, wearing a prescribed and properly fitted compression garment may help decrease swelling during air travel.
Some cases of lower-limb lymphedema have been associated with the use of tamoxifen, due to the blood clots and deep vein thrombosis (DVT) that can be caused by this medication. Resolution of the blood clots or DVT is needed before lymphedema treatment can be initiated.
Congenital lymphedema[edit]
Congenital lymphedema is swelling that results from abnormalities in the lymphatic system that are present from birth. Swelling may be present in a single affected limb, several limbs, genitalia, or the face. It is sometimes diagnosed prenatally by a nuchal scan or post-natally by lymphoscintigraphy. A hereditary form of congenital lymphedema is called Milroy’s disease and is caused by mutations in the VEGFR3 gene.[8] Congenital lymphedema is frequently syndromic and is associated with Turner syndrome, lymphedema–distichiasis syndrome, yellow nail syndrome, and Klippel–Trénaunay–Weber syndrome.[9] In some cases, the condition can sometimes be associated with congenital heart defect, among other things.[10]
Physiology[edit]
Lymph is formed from the fluid that filters out of the blood circulation to nourish cells. This fluid returns through venous capillaries to the blood circulation through the force of osmosis in the venous blood; however, a portion of the fluid that contains proteins, cellular debris, bacteria, etc. must return through the lymphatic collection system to maintain tissue fluid balance. The collection of this prelymph fluid is carried out by the initial lymph collectors that are blind-ended epithelial-lined vessels with fenestrated openings that allow fluids and particles as large as cells to enter. Once inside the lumen of the lymphatic vessels, the fluid is guided along increasingly larger vessels, first with rudimentary valves to prevent backflow, which later develop into complete valves similar to the venous valve. Once the lymph enters the fully valved lymphatic vessels, it is pumped by a rhythmic peristaltic-like action by smooth muscle cells within the lymphatic vessel walls. This peristaltic action is the primary driving force, moving lymph within its vessel walls. The regulation of the frequency and power of contraction is regulated by the sympathetic nervous system. Lymph movement can be influenced by the pressure of nearby muscle contraction, arterial pulse pressure and the vacuum created in the chest cavity during respiration, but these passive forces contribute only a minor percentage of lymph transport. The fluids collected are pumped into continually larger vessels and through lymph nodes, which remove debris and police the fluid for dangerous microbes. The lymph ends its journey in the thoracic duct or right lymphatic duct, which drain into the blood circulation.
Diagnosis[edit]
Assessment of the lower extremities begins with a visual inspection. Color, presence of hair, visible veins, size and any sores or ulcerations are noted. Lack of hair may indicate an arterial circulation problem.[11] Given swelling, the calf circumference is measured for reference as time continues. Elevating the legs may reduce or eliminate the swelling. Palpation of the ankle can determine the degree of swelling. Assessment includes a check of the popliteal, femoral, posterior tibial and dorsalis pedis pulses. The inguinal nodes may be enlarged. Enlargement of the nodes lasting more than three weeks may indicate infection or some other disease process requiring further medical attention.[11]
Diagnosis or early detection of lymphedema is difficult. The first signs may be subjective observations such as “my arm feels heavy” or “I have difficulty these days getting rings on and off my fingers”. These may be symptomatic of early stage of lymphedema where accumulation of lymph is mild and not detectable by changes in volume or circumference. As lymphedema develops further, definitive diagnosis is commonly based upon an objective measurement of differences between the affected or at-risk limb at the opposite unaffected limb, e.g. in volume or circumference. No generally accepted criterion is definitively diagnostic, although a volume difference of 200 ml between limbs or a 4-cm difference (at a single measurement site or set intervals along the limb) is often used. Bioimpedance measurement (which measures the amount of fluid in a limb) offers greater sensitivity than existing methods.[12]
Chronic venous stasis changes can mimic early lymphedema, but the changes in venous stasis are more often bilateral and symmetric. Lipedema can also mimic lymphedema, however lipedema characteristically spares the feet beginning abruptly at the medial malleoli (ankle level). Lipedema is common in overweight women. As a part of the initial work-up before diagnosing lymphedema, it may be necessary to exclude other potential causes of lower extremity swelling such as renal failure, hypoalbuminemia, congestive heart-failure, protein-losing nephropathy, pulmonary hypertension, obesity, pregnancy and drug-induced edema.[13]
Stages[edit]
Whether primary or secondary, lymphedema develops in stages, from mild to severe. Methods of staging are numerous and inconsistent across the globe. Lymphedema staging systems range from three to eight stages.
Staging system of lymphedema to improve diagnosis and outcome[edit]
One staging system was endorsed by the American Society of Lymphology.[14][citation needed] This system provides a four-stage technique that can be employed by clinical and laboratory assessments to more accurately diagnose and prescribe therapy and obtain measurable outcomes. Symptom descriptors and clinical presentation must be established at the assessment by the physician to prescribe interventions, monitor efficacy and support medical necessity. Additional assessments, such as bioimpedance, MRI or CT, build on a clinical assessment (physical evaluation).
The most common method of staging was defined by the Fifth WHO Expert Committee on Filariasis:[15][16]
- Stage 0 (latent): The lymphatic vessels have sustained some damage that is not yet apparent. Transport capacity is sufficient for the amount of lymph being removed. Lymphedema is not present.
- Stage 1 (spontaneously reversible): Tissue is still at the pitting stage: when pressed by the fingertips, the affected area indents and reverses with elevation. Usually upon waking in the morning, the limb or affected area is normal or almost normal in size.
- Stage 2 (spontaneously irreversible): The tissue now has a spongy consistency and is considered non-pitting: when pressed by the fingertips, the affected area bounces back without indentation. Fibrosis found in stage 2 lymphedema marks the beginning of the hardening of the limbs and increasing size.
- Stage 3 (lymphostatic elephantiasis): At this stage, the swelling is irreversible and usually the limb(s) or affected area is noticeably large. The tissue is hard (fibrotic) and unresponsive; some patients consider undergoing reconstructive surgery, called “debulking”. This remains controversial, however, since the risks may outweigh the benefits and the further damage done to the lymphatic system may in fact make the lymphedema worse.
Grades[edit]
Lymphedema can also be categorized by its severity (usually referenced to a healthy extremity):[citation needed]
- Grade 1 (mild edema): Involves the distal parts such as a forearm and hand or a lower leg and foot. The difference in circumference is less than 4 cm and other tissue changes are not yet present.
- Grade 2 (moderate edema): Involves an entire limb or corresponding quadrant of the trunk. Difference in circumference is 4–6 cm. Tissue changes, such as pitting, are apparent. The patient may experience erysipelas.
- Grade 3a (severe edema): Lymphedema is present in one limb and its associated trunk quadrant. Circumferential difference is greater than 6 centimeters. Significant skin alterations, such as cornification or keratosis, cysts and/or fistulae, are present. Additionally, the patient may experience repeated attacks of erysipelas.
- Grade 3b (massive edema): The same symptoms as grade 3a, except that two or more extremities are affected.
- Grade 4 (gigantic edema): Also known as elephantiasis, in this stage of lymphedema, the affected extremities are huge, due to almost complete blockage of the lymph channels. Elephantiasis may also affect the head and face.
Treatment[edit]
Treatment varies depending on edema severity and the degree of fibrosis. Most people with lymphedema follow a daily regimen of treatment. The most common treatments are a combination of manual compression lymphatic massage, compression garments or bandaging. Complex decongestive physiotherapy is an empiric system of lymphatic massage, skin care and compressive garments. Although a combination treatment program may be ideal, any of the treatments can be done individually.
Complete decongestive therapy[edit]
CDT is a primary tool in lymphedema management. It consists of manual manipulation of the lymphatic ducts,[17] short-stretch compression bandaging, therapeutic exercise and skin care. The technique was pioneered by Emil Vodder in the 1930s for the treatment of chronic sinusitis and other immune disorders. Initially, CDT involves frequent visits to a therapist. Once the lymphedema is reduced, increased patient participation is required for ongoing care, along with the use of elastic compression garments and nonelastic directional flow foam garments.
Manual manipulation of the lymphatic ducts (manual lymphatic drainage or MLD) consists of gentle, rhythmic massage to stimulate lymph flow and its return to the blood circulation system. The treatment is gentle. A typical session involves drainage of the neck, trunk and involved extremity (in that order), lasting approximately 40 to 60 minutes. CDT is generally effective on nonfibrotic lymphedema and less effective on more fibrotic legs, although it helps break up fibrotic tissue.[citation needed]
Compression[edit]
Garments[edit]
Elastic compression garments are worn on the affected limb following complete decongestive therapy to maintain edema reduction. Inelastic garments provide containment and reduction.
Bandaging[edit]
Compression bandaging, also called wrapping, is the application of layers of padding and short-stretch bandages to the involved areas. Short-stretch bandages are preferred over long-stretch bandages (such as those normally used to treat sprains), as the long-stretch bandages cannot produce the proper therapeutic tension necessary to safely reduce lymphedema and may in fact end up producing a tourniquet effect. During activity, whether exercise or daily activities, the short-stretch bandages enhance the pumping action of the lymph vessels by providing increased resistance. This encourages lymphatic flow and helps to soften fluid-swollen areas.
A 2002 study showed patients receiving the combined modalities of manual lymph drainage (MLD) with complete decongestive therapy (CDT) and pneumatic pumping had a greater overall reduction in limb volume than patients receiving only MLD/CDT.[18]
Intermittent pneumatic compression therapy[edit]
Intermittent pneumatic compression therapy (IPC) utilizes a multi-chambered pneumatic sleeve with overlapping cells to promote movement of lymph fluid. Pump therapy should be used in addition to other treatments such as compression bandaging and manual lymph drainage. In some cases, pump therapy helps soften fibrotic tissue and therefore potentially enable more efficient lymphatic drainage.[citation needed] However, reports link pump therapy to increased incidence of edema proximal to the affected limb, such as genital edema arising after pump therapy in the lower limb.[19] IPC should be used in combination with complete decongestive therapy.[18]
Exercise[edit]
Most studies investigating the effects exercise in patients with lymphedema or at risk of developing lymphedema examined patients with breast-cancer-related lymphedema. In these studies, resistance training did not increase swelling in patients with pre-existing lymphedema and decreases edema in some patients, in addition to other potential beneficial effects on cardiovascular health.[20][21][22][23] Moreover, resistance training and other forms of exercise were not associated with an increased risk of developing lymphedema in patients who previously received breast cancer-related treatment. Compression garments should be worn during exercise (with the possible exception of swimming in some patients).[24] Patients who have or risk lymphedema should consult their physician or certified lymphedema therapist before beginning an exercise regimen. Resistance training is not recommended in the immediate post-operative period in patients who have undergone axillary lymph node dissection for breast cancer.
Few studies examine the effects of exercise in primary lymphedema or in secondary lymphedema that is not related to breast cancer treatment.
Surgery[edit]
Several surgical procedures provide long-term solutions for patients who suffer from lymphedema. Prior to surgery, patients typically are treated by a physical or occupational therapist, trained in providing lymphedema treatment for initial conservative treatment of their lymphedema. CDT, MLD and compression bandaging are all helpful components of conservative lymphedema treatment.[25]
Vascularized lymph node transfer[edit]
Vascularized lymph node transfers (VLNT) can be an effective treatment of the arm and upper extremity. Lymph nodes are harvested from the groin area with their supporting artery and vein and moved to the axilla (armpit). Microsurgery techniques connect the artery and vein to blood vessels in the axilla to provide support to the lymph nodes while they develop their own blood supply over the first few weeks after surgery.
The newly transferred lymph nodes then serve as a conduit or filter to remove the excess lymphatic fluid from the arm and return it to the body’s natural circulation.
This technique of lymph node transfer usually is performed together with a DIEP flap breast reconstruction. This allows for both the simultaneous treatment of the arm lymphedema and the creation of a breast in one surgery. The lymph node transfer removes the excess lymphatic fluid to return form and function to the arm. In selected cases, the lymph nodes may be transferred as a group with their supporting artery and vein, but without the associated abdominal tissue for breast reconstruction.
Lymph node transfers are most effective in patients whose extremity circumference reduces significantly with compression wrapping, indicating most of the edema is fluid.
VLNT significantly improves the fluid component of lymphedema and decrease the amount of lymphedema therapy and compression garment use required.[26]
Lymphaticovenous anastomosis[edit]
Lymphaticovenous anastomosis (LVA) uses supermicrosurgery to connect the affected lymphatic channels directly to tiny veins located nearby. The lymphatics are tiny, typically 0.1 mm to 0.8 mm in diameter. The procedure requires the use of specialized techniques with superfine surgical suture and an adapted, high-power microscope.
LVA can be an effective and long-term solution for extremity lymphedema and many patients have results that range from a moderate improvement to an almost complete resolution. LVA is most effective early in the course of the disease in patients whose extremity circumference reduces significantly with compression wrapping, indicating most of the edema is fluid. Patients who do not respond to compression are less likely to fare well with LVA, as a greater amount of their increased extremity volume consists of fibrotic tissue, protein or fat. Multiple studies showed LVAs to be effective.[26][27][28]
Lymphaticovenous anastomosis was introduced by B. M. O’Brien and colleagues for the treatment of obstructive lymphedema in the extremities.[29] In 2003, supermicrosurgery pioneer Isao Koshima and colleagues improved the surgery with supermicrosurgical techniques and established the new standard in reconstructive microsurgery.[29] Studies involving long-term follow-up after LVA for lymphedema indicated patients showed remarkable improvement compared to conservative treatment using continuous elastic stocking and occasional pumping.[29]
Clinical studies involving LVA indicate immediate and long-term results showed significant reductions in volume and improvement in systems that appear to be long-lasting.[26][27][30] A 2006 study comparing two groups of breast cancer patients at high risk for lymphedema in whom LVA was used to prevent the onset of clinically evident lymphedema. Results showed a statistically significant reduction in the number of patients who went on to develop clinically significant lymphedema.[30] Other studies showed LVA surgeries reduce the severity of lymphedema in breast cancer patients.[31][32] In particular, a clinical study of 1,000 cases of lymphedema treated with microsurgery from 1973 to 2006 showed beneficial results.[32] Clinical reports from microsurgeons and physical therapists documented more than 1,500 patients treated with LVA surgery over a span of 30 years showing significant improvement and effectiveness.[28]
Indocyanine green fluoroscopy is a safe, minimally invasive and useful tool for surgical evaluation.[33] Microsurgeons use indocyanine green lymphography to assist in LVA surgeries.[34]
Suction assisted lipectomy[edit]
People whose limbs no longer adequately respond to compression therapy may be candidates for suction assisted lipectomy (SAL). This procedure has been called liposuction for lymphedema and is specifically adapted to treat this advanced condition. SAL employs a different operative technique and requires significant therapy and compression garment care that must be administered by a therapist experienced in the technique.
This procedure was pioneered by Hakan Brorson in 1987.[1] Well-controlled clinical trials conducted from 1993 to 2014 showed SAL, combined with controlled compression therapy (CCT), to be an effective lymphedema treatment without recurrence.[1][26][27][35][36][37][38][39] Long-term followup (11–13 years) of patients with lymphedema showed no recurrence of swelling.[1] Lymphatic liposuction combined with controlled compression therapy was more effective than controlled compression therapy alone.[40][41]
SAL has been refined in recent years by using vibrating cannulae that are finer and more effective than previous equipment.[1] In addition, the introduction of the tourniquet and tumescent technique led to minimized blood loss.[1][42]
SAL uses specialized techniques that differ from conventional liposuction procedures and requires specific training.
Lymphatic vessel grafting[edit]
With advanced microsurgical techniques, lymph vessels can be used as grafts. A locally interrupted or obstructed lymphatic pathway, mostly after resection of lymph nodes, can be reconstructed via a bypass using lymphatic vessels. These vessels are specialized to drain lymph by active pumping forces. These grafts are connected with main lymphatic collectors in front and behind the obstruction. The technique is mostly used in arm edemas after treatment of breast cancer and in unilateral edemas of lower extremities after resection of lymph nodes and radiation. The procedure is less widely used than the other surgical procedures, mainly in Germany. The method was developed in 1980 by Ruediger Baumeister.[43]
The method is proven effective.[44] Follow-up studies showed significant volume reduction of the extremities even 10 years after surgery.[45] The patients, who had been previously treated with both MLD and compression therapy, gained significant improvement in quality of life after being treated with lymphatic vessel grafting.[46] Lymphoscintigraphic investigations showed a lasting enhancement of lymphatic transport after grafting.[47]
The patency of lymphatic grafts was demonstrated after more than 12 years, using indirect lymphography and MRI lymphography.
Low level laser therapy[edit]
Low-level laser therapy (LLLT) was cleared by the US Food and Drug Administration (FDA) for the treatment of lymphedema in November 2006.[48]
According to the US National Cancer Institute,
Studies suggest that low-level laser therapy may be effective in reducing lymphedema in a clinically meaningful way for some women. Two cycles of laser treatment were found to be effective in reducing the volume of the affected arm, extracellular fluid, and tissue hardness in approximately one-third of patients with postmastectomy lymphedema at 3 months posttreatment. Suggested rationales for laser therapy include a potential decrease in fibrosis, stimulation of macrophages and the immune system, and a possible role in encouraging lymphangiogenesis.[49][50]
Prevention and disease regression in breast cancer[edit]
In 2008, an NIH study revealed early diagnosis of lymphedema in breast cancer patients (“stage 0”) associated with an early intervention, a compression sleeve and gauntlet for one month, led to a return to preoperative baseline status. In a five-year followup, patients remained at their preoperative baseline, suggesting preclinical detection of lymphedema can halt if not reverse its progression.[citation needed]
Complications[edit]
When the lymphatic impairment becomes so great that the lymph fluid exceeds the lymphatic system’s ability to transport it, an abnormal amount of protein-rich fluid collects in the tissues. Left untreated, this stagnant, protein-rich fluid causes tissue channels to increase in size and number, reducing oxygen availability. This interferes with wound healing and provides a rich culture medium for bacterial growth that can result in infections: cellulitis, lymphangitis, lymphadenitis and in severe cases, skin ulcers. It is vital for lymphedema patients to be aware of the symptoms of infection and to seek immediate treatment, since recurrent infections or cellulitis, in addition to their inherent danger, further damage the lymphatic system and set up a vicious circle.
In rare cases, lymphedema can lead to a form of cancer called lymphangiosarcoma, although the mechanism of carcinogenesis is not understood. Lymphedema-associated lymphangiosarcoma is called Stewart-Treves syndrome. Lymphangiosarcoma most frequently occurs in cases of long-standing lymphedema. The incidence of angiosarcoma is estimated to be 0.45% in patients living 5 years after radical mastectomy.[51][52] Lymphedema is also associated with a low grade form of cancer called retiform hemangioendothelioma (a low grade angiosarcoma).[53]
Since lymphedema is disfiguring, causing difficulties in daily living and can lead to lifestyle becoming severely limited, it may also result in psychological distress.
Manual lymphatic drainage – Wikipedia
Manual lymphatic drainage
Manual lymphatic drainage (MLD) is a type of gentle massage which is intended to encourage the natural drainage of the lymph, which carries waste products away from the tissues back toward the heart. The lymph system depends on intrinsic contractions of the smooth muscle cells in the walls of lymph vessels (peristalsis) and the movement of skeletal muscles to propel lymph through the vessels to lymph nodes and then to the lymph ducts which return lymph to the cardiovascular system. Manual lymph drainage uses a specific amount of pressure (less than 9 ounces per square inch or about 4 kPa) and rhythmic circular movements to stimulate lymph flow.[1][2]
History[edit]
Manual lymphatic drainage was pioneered by Danish Drs. Emil Vodder and Estrid Vodder in the 1930s[2] for the treatment of chronic sinusitis and other immune disorders. While working on the French Riviera treating patients with chronic colds, the Vodders noticed these patients had swollen lymph nodes. In the 1930s, the lymphatic system was poorly understood. The Vodders were not deterred by this and, in 1932, began to study the lymph system, developing light, rhythmic hand movements to promote lymph movement. In 1936, they introduced this technique in Paris, France, and after World War II, they returned to Copenhagen to teach other practitioners to use this therapy.[3]
Recognition[edit]
MLD is now recognized as a primary tool in lymphedema management. Therapists can today receive certification through special classes conducted by various organizations specializing in MLD, or through a complete lymphedema treatment certification course.[4] Scientific studies show mixed results regarding the efficacy of the method in treating lymphedema and further studies are needed.[5] A 2009 meta-analysis of studies in the area of sports medicine and rehabilitation showed best evidence of effectiveness for Manual lymphatic drainage treatment to “enzyme serum levels associated with acute skeletal muscle cell damage as well as reduction of edema [swelling] around broken bones.” [6] A 2013 systematic review of manual lymphatic drainage with regard to breast cancer related lymphedema found no clear support for the effectiveness of the intervention in either preventing limb edema in at-risk women or treating women for the condition.[7]
Lymphatic system – Wikipedia
Lymphatic system
| Lymphatic System | |
|---|---|
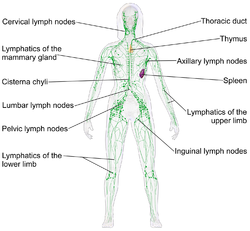
Human lymphatic system
|
|
 |
|
| Details | |
| Identifiers | |
| Latin | systema lymphoideum |
| TA | 13.0.00.000 |
| FMA | 74594 |
| Anatomical terminology | |
The lymphatic system is part of the circulatory system and a vital part of the immune system, comprising a network of lymphatic vessels that carry a clear fluid called lymph (from Latin, lympha meaning water[1]) directionally towards the heart. The lymphatic system was first described in the seventeenth century independently by Olaus Rudbeck and Thomas Bartholin. Unlike the cardiovascular system, the lymphatic system is not a closed system. The human circulatory system processes an average of 20 liters of blood per day through capillary filtration, which removes plasma while leaving the blood cells. Roughly 17 litres of the filtered plasma are reabsorbed directly into the blood vessels, while the remaining three litres remain in the interstitial fluid. One of the main functions of the lymph system is to provide an accessory return route to the blood for the surplus three litres.[2]
The other main function is that of defense in the immune system. Lymph is very similar to blood plasma: it contains lymphocytes and other white blood cells. It also contains waste products and cellular debris together with bacteria and proteins. Associated organs composed of lymphoid tissue are the sites of lymphocyte production. Lymphocytes are concentrated in the lymph nodes. The spleen and the thymus are also lymphoid organs of the immune system. The tonsils are lymphoid organs that are also associated with the digestive system. Lymphoid tissues contain lymphocytes, and also contain other types of cells for support.[3] The system also includes all the structures dedicated to the circulation and production of lymphocytes (the primary cellular component of lymph), which also includes the bone marrow, and the lymphoid tissue associated with the digestive system.[4]
The blood does not come into direct contact with the parenchymal cells and tissues in the body (except in case of an injury causing rupture of one or more blood vessels), but constituents of the blood first exit the microvascular exchange blood vessels to become interstitial fluid, which comes into contact with the parenchymal cells of the body. Lymph is the fluid that is formed when interstitial fluid enters the initial lymphatic vessels of the lymphatic system. The lymph is then moved along the lymphatic vessel network by either intrinsic contractions of the lymphatic passages or by extrinsic compression of the lymphatic vessels via external tissue forces (e.g., the contractions of skeletal muscles), or by lymph hearts in some animals. The organization of lymph nodes and drainage follows the organization of the body into external and internal regions; therefore, the lymphatic drainage of the head, limbs, and body cavity walls follows an external route, and the lymphatic drainage of the thorax, abdomen, and pelvic cavities follows an internal route.[5] Eventually, the lymph vessels empty into the lymphatic ducts, which drain into one of the two subclavian veins, near their junction with the internal jugular veins.
Structure[edit]
The lymphatic system consists of lymphatic organs, a conducting network of lymphatic vessels, and the circulating lymph.
The thymus and the bone marrow constitute the primary lymphoid organs involved in the production and early clonal selection of lymphocyte tissues. Bone marrow is responsible for both the creation of T cells and the production and maturation of B cells. From the bone marrow, B cells immediately join the circulatory system and travel to secondary lymphoid organs in search of pathogens. T cells, on the other hand, travel from the bone marrow to the thymus, where they develop further. Mature T cells join B cells in search of pathogens. The other 95% of T cells begin a process of apoptosis, a form of programmed cell death.
The central or primary lymphoid organs generate lymphocytes from immature progenitor cells.
Secondary or peripheral lymphoid organs, which include lymph nodes and the spleen, maintain mature naive lymphocytes and initiate an adaptive immune response. The peripheral lymphoid organs are the sites of lymphocyte activation by antigens. Activation leads to clonal expansion and affinity maturation. Mature lymphocytes recirculate between the blood and the peripheral lymphoid organs until they encounter their specific antigen.
Secondary lymphoid tissue provides the environment for the foreign or altered native molecules (antigens) to interact with the lymphocytes. It is exemplified by the lymph nodes, and the lymphoid follicles in tonsils, Peyer’s patches, spleen, adenoids, skin, etc. that are associated with the mucosa-associated lymphoid tissue (MALT).
In the gastrointestinal wall the appendix has mucosa resembling that of the colon, but here it is heavily infiltrated with lymphocytes.
The tertiary lymphoid tissue typically contains far fewer lymphocytes, and assumes an immune role only when challenged with antigens that result in inflammation. It achieves this by importing the lymphocytes from blood and lymph.[6])
Lymphoid tissue[edit]
Thymus[edit]
The thymus is a primary lymphoid organ and the site of maturation for T cells, the lymphocytes of the adaptive immune system. The thymus increases in size from birth in response to postnatal antigen stimulation, then to puberty and regresses thereafter.[7] The loss or lack of the thymus results in severe immunodeficiency and subsequent high susceptibility to infection.[7] In most species, the thymus consists of lobules divided by septa which are made up of epithelium and is therefore an epithelial organ. T cells mature from thymocytes, proliferate and undergo selection process in the thymic cortex before entering the medulla to interact with epithelial cells.
The thymus provides an inductive environment for development of T cells from hematopoietic progenitor cells. In addition, thymic stromal cells allow for the selection of a functional and self-tolerant T cell repertoire. Therefore, one of the most important roles of the thymus is the induction of central tolerance.
The thymus is largest and most active during the neonatal and pre-adolescent periods. By the early teens, the thymus begins to atrophy and thymic stroma is mostly replaced by adipose tissue. Nevertheless, residual T lymphopoiesis continues throughout adult life.
Spleen[edit]
The main functions of the spleen are:
- to produce immune response against blood-borne antigens
- to remove particulate matter and aged blood cells, mainly erythrocytes
- to produce blood cells during fetal life
The spleen synthesizes antibodies in its white pulp and removes antibody-coated bacteria and antibody-coated blood cells by way of blood and lymph node circulation. A study published in 2009 using mice found that the spleen contains, in its reserve, half of the body’s monocytes within the red pulp.[8] These monocytes, upon moving to injured tissue (such as the heart), turn into dendritic cells and macrophages while promoting tissue healing.[8][9][10] The spleen is a center of activity of the mononuclear phagocyte system and can be considered analogous to a large lymph node, as its absence causes a predisposition to certain infections.
Like the thymus, the spleen has only efferent lymphatic vessels. Both the short gastric arteries and the splenic artery supply it with blood.[11]
The germinal centers are supplied by arterioles called penicilliary radicles.[12]
Up to the fifth month of prenatal development the spleen creates red blood cells. After birth the bone marrow is solely responsible for hematopoiesis. As a major lymphoid organ and a central player in the reticuloendothelial system, the spleen retains the ability to produce lymphocytes. The spleen stores red blood cells and lymphocytes. It can store enough blood cells to help in an emergency. Up to 25% of lymphocytes can be stored at any one time.[13]
Lymph nodes[edit]
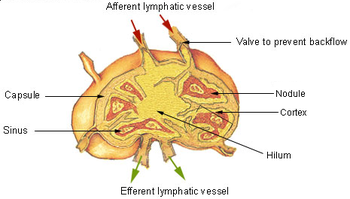
A lymph node showing afferent and efferent lymphatic vessels
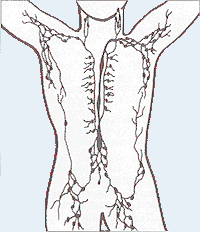
A lymph node is an organized collection of lymphoid tissue, through which the lymph passes on its way back to the blood. Lymph nodes are located at intervals along the lymphatic system. Several afferent lymph vessels bring in lymph, which percolates through the substance of the lymph node, and is then drained out by an efferent lymph vessel. There are between five and six hundred lymph nodes in the human body, many of which are grouped in clusters in different regions as in the underarm and abdominal areas. Lymph node clusters are commonly found at the base of limbs (groin, armpits) and in the neck, where lymph is collected from regions of the body likely to sustain pathogen contamination from injuries.
The substance of a lymph node consists of lymphoid follicles in an outer portion called the cortex. The inner portion of the node is called the medulla, which is surrounded by the cortex on all sides except for a portion known as the hilum. The hilum presents as a depression on the surface of the lymph node, causing the otherwise spherical lymph node to be bean-shaped or ovoid. The efferent lymph vessel directly emerges from the lymph node at the hilum. The arteries and veins supplying the lymph node with blood enter and exit through the hilum.
The region of the lymph node called the paracortex immediately surrounds the medulla. Unlike the cortex, which has mostly immature T cells, or thymocytes, the paracortex has a mixture of immature and mature T cells. Lymphocytes enter the lymph nodes through specialised high endothelial venules found in the paracortex.
A lymph follicle is a dense collection of lymphocytes, the number, size and configuration of which change in accordance with the functional state of the lymph node. For example, the follicles expand significantly when encountering a foreign antigen. The selection of B cells, or B lymphocytes, occurs in the germinal center of the lymph nodes.
Lymph nodes are particularly numerous in the mediastinum in the chest, neck, pelvis, axilla, inguinal region, and in association with the blood vessels of the intestines.[4]
Other lymphoid tissue[edit]
Lymphoid tissue associated with the lymphatic system is concerned with immune functions in defending the body against infections and the spread of tumors. It consists of connective tissue formed of reticular fibers, with various types of leukocytes, (white blood cells), mostly lymphocytes enmeshed in it, through which the lymph passes.[14] Regions of the lymphoid tissue that are densely packed with lymphocytes are known as lymphoid follicles. Lymphoid tissue can either be structurally well organized as lymph nodes or may consist of loosely organized lymphoid follicles known as the mucosa-associated lymphoid tissue.
The central nervous system also has lymphatic vessels, as discovered by University of Virginia Researchers. The search for T-cell gateways into and out of the meninges uncovered functional lymphatic vessels lining the dural sinuses, anatomically integrated into the membrane surrounding the brain.[15]
Lymphatics[edit]
The lymphatic vessels, also called lymph vessels, conduct lymph between different parts of the body. They include the tubular vessels of the lymph capillaries, and the larger collecting vessels–the right lymphatic duct and the thoracic duct (the left lymphatic duct). The lymph capillaries are mainly responsible for the absorption of interstitial fluid from the tissues, while lymph vessels propel the absorbed fluid forward into the larger collecting ducts, where it ultimately returns to the bloodstream via one of the subclavian veins. These vessels are also called the lymphatic channels or simply lymphatics.[16]
The lymphatics are responsible for maintaining the balance of the body fluids. Its network of capillaries and collecting lymphatic vessels work to efficiently drain and transport extravasated fluid, along with proteins and antigens, back to the circulatory system. Numerous intraluminal valves in the vessels ensure a unidirectional flow of lymph without reflux.[17] Two valve systems are used to achieve this one directional flow—a primary and a secondary valve system.[18] The capillaries are blind-ended, and the valves at the ends of capillaries use specialised junctions together with anchoring filaments to allow a unidirectional flow to the primary vessels. The collecting lymphatics, however, act to propel the lymph by the combined actions of the intraluminal valves and lymphatic muscle cells.[19]
Development[edit]
Lymphatic tissues begin to develop by the end of the fifth week of embryonic development. Lymphatic vessels develop from lymph sacs that arise from developing veins, which are derived from mesoderm.
The first lymph sacs to appear are the paired jugular lymph sacs at the junction of the internal jugular and subclavian veins. From the jugular lymph sacs, lymphatic capillary plexuses spread to the thorax, upper limbs, neck and head. Some of the plexuses enlarge and form lymphatic vessels in their respective regions. Each jugular lymph sac retains at least one connection with its jugular vein, the left one developing into the superior portion of the thoracic duct.
The next lymph sac to appear is the unpaired retroperitoneal lymph sac at the root of the mesentery of the intestine. It develops from the primitive vena cava and mesonephric veins. Capillary plexuses and lymphatic vessels spread from the retroperitoneal lymph sac to the abdominal viscera and diaphragm. The sac establishes connections with the cisterna chyli but loses its connections with neighboring veins.
The last of the lymph sacs, the paired posterior lymph sacs, develop from the iliac veins. The posterior lymph sacs produce capillary plexuses and lymphatic vessels of the abdominal wall, pelvic region, and lower limbs. The posterior lymph sacs join the cisterna chyli and lose their connections with adjacent veins.
With the exception of the anterior part of the sac from which the cisterna chyli develops, all lymph sacs become invaded by mesenchymal cells and are converted into groups of lymph nodes.
The spleen develops from mesenchymal cells between layers of the dorsal mesentery of the stomach. The thymus arises as an outgrowth of the third pharyngeal pouch.
Function[edit]
The lymphatic system has multiple interrelated functions:[20]
- It is responsible for the removal of interstitial fluid from tissues
- It absorbs and transports fatty acids and fats as chyle from the digestive system
- It transports white blood cells to and from the lymph nodes into the bones
- The lymph transports antigen-presenting cells, such as dendritic cells, to the lymph nodes where an immune response is stimulated.
Function of the fatty acid transport system[edit]
Lymph vessels called lacteals are present in the lining of the gastrointestinal tract, predominantly in the small intestine. While most other nutrients absorbed by the small intestine are passed on to the portal venous system to drain via the portal vein into the liver for processing, fats (lipids) are passed on to the lymphatic system to be transported to the blood circulation via the thoracic duct. (There are exceptions, for example medium-chain triglycerides are fatty acid esters of glycerol that passively diffuse from the GI tract to the portal system.) The enriched lymph originating in the lymphatics of the small intestine is called chyle. The nutrients that are released to the circulatory system are processed by the liver, having passed through the systemic circulation.
Immune function[edit]
The lymphatic system plays a major role in body’s immune system, as the primary site for cells relating to adaptive immune system including T-cells and B-cells. Cells in the lymphatic system react to antigens presented or found by the cells directly or by other dendritic cells. When an antigen is recognised, an immunological cascade begins involving the activation and recruitment of more and more cells, the production of antibodies and cytokines and the recruitment of other immunological cells such as macrophages.
Clinical significance[edit]
The study of lymphatic drainage of various organs is important in the diagnosis, prognosis, and treatment of cancer. The lymphatic system, because of its closeness to many tissues of the body, is responsible for carrying cancerous cells between the various parts of the body in a process called metastasis. The intervening lymph nodes can trap the cancer cells. If they are not successful in destroying the cancer cells the nodes may become sites of secondary tumors.
Lymphadenopathy[edit]
Lymphadenopathy refers to one or more enlarged lymph nodes. Small groups or individually enlarged lymph nodes are generally reactive in response to infection or inflammation. This is called local lymphadenopathy. When many lymph nodes in different areas of the body are involved, this is called generalised lymphadenopathy. Generalised lymphadenopathy may be caused by infections such as infectious mononucleosis, tuberculosis and HIV, connective tissue diseases such as SLE and rheumatoid arthritis, and cancers, including both cancers of tissue within lymph nodes, discussed below, and metastasis of cancerous cells from other parts of the body, that have arrived via the lymphatic system.[21]
Lymphedema[edit]
Lymphedema is the swelling caused by the accumulation of lymph, which may occur if the lymphatic system is damaged or has malformations. It usually affects limbs, though the face, neck and abdomen may also be affected. In an extreme state, called elephantiasis, the edema progresses to the extent that the skin becomes thick with an appearance similar to the skin on elephant limbs.[22]
Causes are unknown in most cases, but sometimes there is a previous history of severe infection, usually caused by a parasitic disease, such as lymphatic filariasis.
Lymphangiomatosis is a disease involving multiple cysts or lesions formed from lymphatic vessels.
Lymphedema can also occur after surgical removal of cancerous lymph nodes in the armpit (causing the arm to swell due to poor lymphatic drainage) or groin (causing swelling of the leg). Treatment is by massage, and is not permanent.
Cancer[edit]
Cancer of the lymphatic system can be primary or secondary. Lymphoma refers to cancer that arises from lymphatic tissue. Lymphoid leukemias and lymphomas are now considered to be tumors of the same type of cell lineage. They are called “leukemia” when in the blood or marrow and “lymphoma” when in lymphatic tissue. They are grouped together under the name “lymphoid malignancy”.[23]
Lymphoma is generally considered as either Hodgkin lymphoma or non-Hodgkin lymphoma. Hodgkin lymphoma is characterised by a particular type of cell, called a Reed–Sternberg cell, visible under microscope. It is associated with past infection with the Epstein-Barr Virus, and generally causes a painless “rubbery” lymphadenopathy. It is staged, using Ann Arbor staging. Chemotherapy generally involves the ABVD and may also involve radiotherapy.[24] Non-Hodgkin lymphoma is a cancer characterised by increased proliferation of B-cells or T-cells, generally occurs in an older age group than Hodgkin lymphoma. It is treated according to whether it is high-grade or low-grade, and carries a poorer prognosis than Hodgkin lymphoma.[24]
Lymphangiosarcoma is a malignant soft tissue tumor, whereas lymphangioma is a benign tumor occurring frequently in association with Turner syndrome. Lymphangioleiomyomatosis is a benign tumor of the smooth muscles of the lymphatics that occurs in the lungs.
Other[edit]
- Lymphangitis
- Kikuchi disease
- Chylothorax
- Castleman’s disease
- Lymphatic filariasis
- Solitary lymphatic nodule
History[edit]
Hippocrates, in 5th century BC, was one of the first people to mention the lymphatic system. In his work On Joints, he briefly mentioned the lymph nodes in one sentence.